Cryopreservation: How Good is Good Enough?
Why cryopreservation is difficult, and CT scans are not sufficient to show quality
I recently visited some of the people working at Nectome in Portland, OR. I learned a lot about cryopreservation quality from them. I try to fact-check somewhat, but I am not an expert in this area. (I may edit this in the next few days with feedback from people)
I will discuss some about blood reflow, and how this affects perfusion quality, and also explain how CT scans are probably not good enough to determine quality.
How good is good enough?
In short, we don’t know what future technology looks like, or how information is stored in neurons, or what causes it to be destroyed or not.
It might be that you need really high preservation quality or the information to be usabale, perhaps at or exceeding current standards in neuroscience. Or, it might be easy to reconstruct the original state after the damage with some future nano-tech or AI or something. Nobody really knows.
Given uncertainty, it seems better to aim for as high a quality of preservation as possible, but what is that according to current standards?
Perfusion
The key desiderata, are that we want to keep the cells in a state that is as close to the original as possible. If one were to freeze the cells normally, this would typically lead to ice crystals destroying the cells and leaving them quite damaged. Is this fine? Nobody knows. But it seems much worse to allow this to happen, than to prevent the cells from being damaged.
As I discussed in “Two Theories for Cryopreservation”, there are two main ways this can be done:
Vitrification, which replacing the water with a fluid that doesn’t form crystals the way ice does
Aldehyde Fixation: essentially turning the cells into a plastic-like state by cross-linking all of the cell pieces chemically.
Both have pros and cons, but both only work if one can get the fluid to the cells that need to be preserved, especially the neurons in our brain. This mostly involves pumping the cryoprotective agents though the arteries in our existing circulatory system, and hoping that is sufficiently in-tact that it gets to all of the cells.
So how viable is that?
No-reflow phenomenon
If we look at the brain, we can see that, like most of the body, it has some main arteries, which branch off into smaller arteries, which then branch of into many capillaries flow blood through the brain and which are used to ensure our neurons get a continuous supply of oxygen.
Capillaries are incredibly small, such that red blood cells need to physically deform to pass through them. This means that any loss in size of capillary diameter makes it difficult-to-impossible to get blood flowing through them.
When your heart stops, your blood stops moving, and your cells quickly being to respond to the lack of oxygen, often leading to changes to the size of your blood vessels. The different sizes alter the dynamics of how much blood flows through different parts of your body.
And so, a key challenge is with trying to get cryoprotective agents through your circulatory system by getting your blood moving again.
There are many factors that mean an ischemic brain kills itself. Claude tells me there are like 30-40 different factors1 and there is a large literature on this. And the only known way to reliably affect all of these is to have a lower temperature.2
An important way this matters to us, is that ischemia causes capillaries to contract and become smaller. There is a time window where it is possible to reach the whole brain, after which the capillaries become too small such that pumping blood or cryoprotective agents through the body just doesn’t reach most places.
capillary constriction at pericyte locations was seen in coronary no reflow in a rat model of myocardial ischemia and reperfusion. They noted that 40% of capillaries had no reflow at pericyte locations, where capillary diameter was reduced by 37%.
This ~60% decrease in cross-area thus significantly constraints cryopreservation quality
Can we see whether the preservation quality was good?
We can use tools to try probe whether perfusion of cryoprotective agents into all of the human neurons was successful.
The first way we can try to do this, is via a CT scan.
One can look at the density of various parts of the brain, and see whether there are large areas and look at denisty to see whether the cryoprotective agent has successfuly diffused to all parts of the brain. This only has a resolution of about ~0.5mm so can only give a rough area, but as mentioned in recent work by “As far as preservation quality goes, good CT scans are a necessary condition, but not a sufficient one”
Neuron sizes are much smaller than 0.5mm. According to this nature article: “The synaptic cleft represents the intercellular space of approximately 20–30 nm” and so we need a much much finer resolution to determine whether the micro-structure of the neurons is in-tact.
Another method one can try is light-based microscopy.
Light-based microscopy tools can be used to see supplement seeing how well smaller structures are cryopreserved, but the resolution for this is diffraction-limited to ~200nm. One can use this as another necessary condition for assessing quality, but yet again, it is not sufficient.
Lastly, we can use the most expensive method, which is electron microscopy.
This has a pixel size of more like 5-10nm, and so for the brain preservation foundation prize, one explains that for their testing “sufficient number of high-resolution (5nm pixel size) micrographs will be taken to provide a convincing case that the entire surface shows good ultrastructure preservation.”
So how well do current method work?
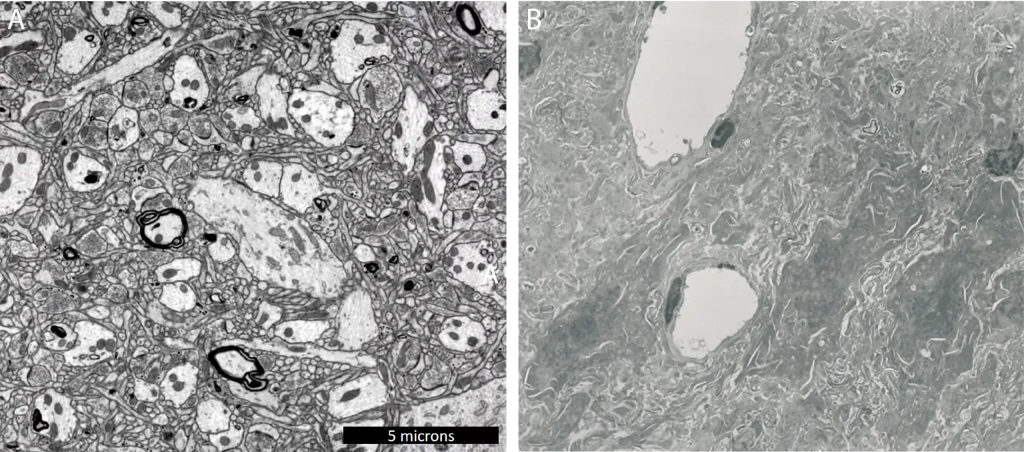
My understanding from the conversations with Nectome, is that existing methods for cryopreservation can often give results that look good with both CT scans and light-based microscopy methods, but when looking at neurons using electron microscopy, one can start to see issues more clearly.
Using aldehyde based-methods, Nectome recently established “14 min is the approximate length of the perfusability window”. Beyond this, one may get reasonable light microscopy results, but stochastically whole parts of the brain may not be correctly cryopreserved.
One can look at recent research by Nectome and see a visual depiction of how these differ:
Going Forward
I agree it still seems worthwhile to try save people who have died suddenly, even if preservation quality might be affected. However, it seems valuable that Nectome is trying to create a reference best-case quality preservation in best-case scenarios too.
I can understand reservations about making aldehyde fixation making brain uploading more likely than biological revival (I wrote a whole post about this, as well discussed some about continuity of consciousness), but it seems unclear if traditional vitrification is really more likely to lead to biological revival either.
I hope that Tomorrow bio is successful with releasing their micro-structure results in the coming year, based on information from Nectome, my understanding is that there is a high chance the results will come back negative.
According to Claude:
There exists literature describing 30–40 partially independent mechanisms by which an ischemic brain kills itself: ATP depletion, Na⁺/K⁺-ATPase failure, glutamate excitotoxicity, NMDA/AMPA-receptor overactivation, calcium overload, ROS production, lipid peroxidation, mitochondrial permeability transition, ferroptosis, necroptosis, PANoptosis, calpain cleavage of cytoskeleton, lysosomal permeabilization, BBB tight-junction degradation, cytotoxic edema, vasogenic edema, pericyte constriction, neutrophil plugging, microthrombi, spreading depolarizations... (Qin et al. 2022, Nature STTT; Siesjö 1988).
Decades of trials targeting single mechanisms — NMDA antagonists, free-radical scavengers, Ca²⁺ blockers, magnesium — have mostly failed. The only broad-spectrum intervention that works is hypothermia, because it slows all of it at once.
One may think that cooling the skin with ice would work, but this is too slow. You would need to pump the cold through the person via their circulatory system, either directly though blood or by cooling it through their lungs.
Mechanisms of the “No-Reflow” Phenomenon After Acute Myocardial Infarction Potential Role of Pericytes - Kaul et al. 2022



https://www.lesswrong.com/posts/3i5GMhpGbDwef9Rns/nectome-all-that-i-know?commentId=DztqNMQjjrQummzSk